After President Trump was criticized for suggesting “[bringing] the light inside the body, which you can do either through the skin or in some other way” to kill coronavirus, many on Twitter pointed to an ultraviolet treatment proposed by pharmaceutical company Aytu BioScience as evidence that Trump’s idea could be accomplished:
The tweets contained a link to a YouTube video that has since been removed from the service. Some pointed to the removal of a Aytu BioScience video explaining the treatment from YouTube and the suspension of the company’s Twitter account as “censorship” intended to make the president look bad:
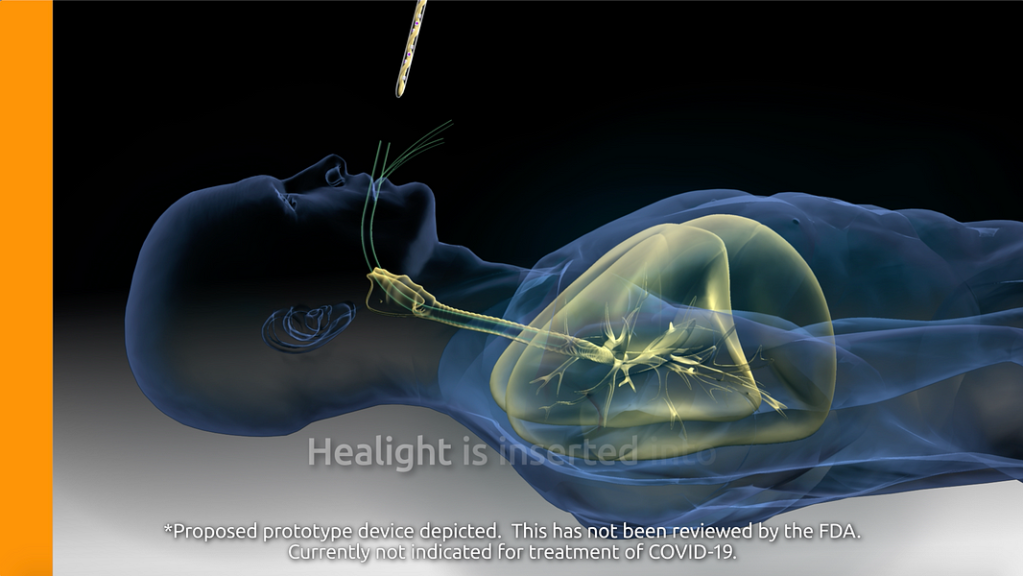
Aytu BioScience’s website states that the basis for its proposed prototype, named “Healight,” is a 2019 study titled “Internally Applied Ultraviolet Light as a Novel Approach for Effective and Safe Anti-Microbial Treatment,” published in the United European Gastroenterology Journal. The article isn’t accessible without an account (and our attempts to create an account resulted in error messages), but Aytu BioScience’s Healight web page summarizes the findings: “Here, the authors show that UVA light exhibits significant in vitro bactericidal effects in an array of clinically important bacteria. Additionally, this is the first study using intracolonic UVA application, which reports that UVA exposure is not associated with endoscopic or histologic injury.”
Aytu BioScience’s proposal for a UV light treatment has not yet been fully tested. The company’s website warns that “[Healight] has not been reviewed by the FDA” and it warns specifically that it is not approved for use in the fight against coronavirus. “This device, or concept of this device is currently not indicated for use in the treatment of COVID-19.” The video that was removed from YouTube can still be found on AYTU BioScience’s website, and it bears a similar disclaimer: “Proposed prototype device depicted. This has not been reviewed by the FDA. Currently not indicated for treatment of COVID-19.” Aytu BioScience CEO Josh Disbrow wrote an op-ed for The Wall Street Journal about the politicization of his company’s research, in which he expressed hope that the new technology could eventually be used on coronavirus but conceded, “There is still much work to do on Healight—much to study, and much to verify.”
Some experts are skeptical that even after the work is done Healight will amount to much. There’s an important distinction to be made between forms of ultraviolet light. Ultraviolet light exists on a spectrum split up by wavelength; ultraviolet A (UVA) light has the longest wavelengths, followed by ultraviolet B (UVB) light, while ultraviolet C (UVA) light has the shortest. Aytu BioScience’s Healight uses UVA light. Another form of ultraviolet light that’s being explored to combat coronavirus, though to kill the virus in the air not in humans, is a specific, very short form of UVC light called far-UVC light.
Dr. David Brenner, director of Columbia University’s Center for Radiological Research, has been exploring ultraviolet technology to combat coronavirus, and he has seen promise using far-UVC light. He doubts UVA light would work and thinks it may even be dangerous to internal tissue.
“It’s an interesting idea, though I have doubts that it would work,” Brenner told the Dispatch Fact Check. “The airways and lung are pretty complex geometrical structures, so any small amount of material between the virus and the UV light source would protect the virus.”
“In addition, UVA is not particularly effective at killing viruses—far less effective than UVC,” Brenner said. He mentioned one 2006 paper from the FDA’s Center for Biologics Evaluation and Research that showed while UVC light efficiently kills SAR-CoV-1 (the virus that caused the 2003 outbreak of SARS), UVA light does not.
“Finally, I have some concerns about safety of the lining of lung exposed directly to the UVA light, without any protective layer of dead cells, which is typically the case with external exposure to sunlight,” Brenner said.
This isn’t the first time the Aytu BioScience has faced questions about its products. Earlier this year the company made news for selling coronavirus tests produced by Chinese manufacturers whose tests did not meet Chinese regulatory standards. Aytu’s website notes: “The FDA has not reviewed Aytu BioScience’s validation of the [coronavirus] test.” The Denver Public Safety Department used Aytu’s tests, and a spokeswoman said the tests generated an unexpectedly high number of false positives.
Was the content removed simply because it was touted by Donald Trump? The company acknowledges in both the written and video content itself that it is not FDA-approved and is not “indicated for treatment of COVID-19.” YouTube CEO Susan Wojcicki said in an interview that the platform would remove any “medically unsubstantiated” videos about coronavirus and Twitter leadership has similarly announced it will remove anything that “goes directly against guidance from authoritative sources of global and local public health information.” While there is an ongoing debate about whether those guidelines are the right ones, Healight meets the stated criteria for removal from both websites.
If you have a claim you would like to see us fact check, please send us an email at factcheck@thedispatch.com. If you would like to suggest a correction to this piece or any other Dispatch article, please email corrections@thedispatch.com.








Please note that we at The Dispatch hold ourselves, our work, and our commenters to a higher standard than other places on the internet. We welcome comments that foster genuine debate or discussion—including comments critical of us or our work—but responses that include ad hominem attacks on fellow Dispatch members or are intended to stoke fear and anger may be moderated.
You are currently using a limited time guest pass and do not have access to commenting. Consider subscribing to join the conversation.
With your membership, you only have the ability to comment on The Morning Dispatch articles. Consider upgrading to join the conversation everywhere.